How Many Protons In Beryllium
 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Beryllium | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pronunciation | | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Advent | white-gray metallic | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Standard atomic weight A r°(Exist) |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Glucinium in the periodic table | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic number (Z) | four | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Group | group two (alkaline metal earth metals) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Period | period 2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Block | due south-cake | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electron configuration | [He] 2stwo | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electrons per shell | 2, 2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Phase atSTP | solid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting betoken | 1560 K (1287 °C, 2349 °F) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Boiling bespeak | 2742 K (2469 °C, 4476 °F) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density (nearr.t.) | one.85 g/cmthree | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| when liquid (atm.p.) | 1.690 g/cm3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Critical bespeak | 5205 K, MPa (extrapolated) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Heat of fusion | 12.2 kJ/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Rut of vaporization | 292 kJ/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molar heat capacity | xvi.443 J/(mol·G) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Vapor pressure
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Oxidation states | 0,[2] +ane,[3] +two (an amphoteric oxide) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electronegativity | Pauling scale: 1.57 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ionization energies |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Diminutive radius | empirical: 112 pm | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Covalent radius | 96±three pm | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Van der Waals radius | 153 pm | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Natural occurrence | primordial | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Crystal structure | hexagonal shut-packed (hcp) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Speed of audio thin rod | 12,890 grand/s (atr.t.)[4] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Thermal expansion | eleven.3 µm/(m⋅K) (at 25 °C) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Thermal conductivity | 200 W/(m⋅G) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electrical resistivity | 36 nΩ⋅m (at 20 °C) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Magnetic ordering | diamagnetic | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molar magnetic susceptibility | −9.0×x−six cm3/mol[5] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Immature's modulus | 287 GPa | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Shear modulus | 132 GPa | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Bulk modulus | 130 GPa | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Poisson ratio | 0.032 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mohs hardness | 5.v | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Vickers hardness | 1670 MPa | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Brinell hardness | 590–1320 MPa | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS Number | 7440-41-7 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| History | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Discovery | Louis Nicolas Vauquelin (1798) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| First isolation | Friedrich Wöhler & Antoine Bussy (1828) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Main isotopes of glucinium | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Beryllium is a element with the symbol Be and atomic number four. It is a steel-grey, strong, lightweight and breakable element of group ii. It is a divalent element that occurs naturally only in combination with other elements to course minerals. Notable gemstones high in beryllium include beryl (aquamarine, emerald) and chrysoberyl. Information technology is a relatively rare element in the universe, ordinarily occurring every bit a product of the spallation of larger atomic nuclei that take collided with catholic rays. Inside the cores of stars, glucinium is depleted every bit it is fused into heavier elements. Glucinium constitutes well-nigh 0.0004 percent by mass of Earth's chaff. The world'due south almanac beryllium production of 220 tons is usually manufactured by extraction from the mineral beryl, a difficult process because beryllium bonds strongly to oxygen.
In structural applications, the combination of high flexural rigidity, thermal stability, thermal conductivity and low density (1.85 times that of water) make beryllium metallic a desirable aerospace material for aircraft components, missiles, spacecraft, and satellites.[half dozen] Because of its low density and atomic mass, beryllium is relatively transparent to X-rays and other forms of ionizing radiation; therefore, it is the most common window material for X-ray equipment and components of particle detectors.[half-dozen] When added equally an alloying chemical element to aluminium, copper (notably the alloy glucinium copper), iron, or nickel, beryllium improves many physical properties.[6] For instance, tools and components made of glucinium copper alloys are strong and hard and do non create sparks when they strike a steel surface. In air, the surface of glucinium oxidizes readily at room temperature to course a passivation layer 1–10 nm thick that protects information technology from further oxidation and corrosion. The metal oxidizes in bulk (beyond the passivation layer) when heated in a higher place 500 °C (932 °F), and burns brilliantly when heated to near ii,500 °C (4,530 °F).
The commercial use of beryllium requires the use of appropriate dust control equipment and industrial controls at all times considering of the toxicity of inhaled glucinium-containing dusts that can cause a chronic life-threatening allergic disease in some people called berylliosis.[7] Berylliosis causes pneumonia and other associated respiratory illness.
Characteristics [edit]
Physical properties [edit]
Beryllium is a steel gray and difficult metallic that is breakable at room temperature and has a shut-packed hexagonal crystal structure.[6] It has exceptional stiffness (Young'due south modulus 287 GPa) and a melting point of 1287 °C. The modulus of elasticity of beryllium is approximately 50% greater than that of steel. The combination of this modulus and a relatively low density results in an unusually fast sound conduction speed in beryllium – about 12.9 km/s at ambient conditions. Other significant properties are loftier specific heat ( 1925 J·kg−1·K−1 ) and thermal conductivity ( 216 W·k−ane·K−1 ), which make glucinium the metal with the all-time heat dissipation characteristics per unit weight. In combination with the relatively low coefficient of linear thermal expansion (xi.4×ten−6 1000−1), these characteristics outcome in a unique stability under weather of thermal loading.[8]
Nuclear properties [edit]
Naturally occurring beryllium, save for slight contamination past the cosmogenic radioisotopes, is isotopically pure beryllium-9, which has a nuclear spin of three / ii . Glucinium has a large handful cross department for loftier-energy neutrons, almost half-dozen barns for energies above approximately 10 keV. Therefore, it works every bit a neutron reflector and neutron moderator, effectively slowing the neutrons to the thermal energy range of below 0.03 eV, where the total cross section is at least an club of magnitude lower; the exact value strongly depends on the purity and size of the crystallites in the textile.
The single primordial beryllium isotope 9Be besides undergoes a (north,2n) neutron reaction with neutron energies over nigh 1.ix MeV, to produce 8Be, which almost immediately breaks into two alpha particles. Thus, for high-energy neutrons, beryllium is a neutron multiplier, releasing more neutrons than information technology absorbs. This nuclear reaction is:[9]
- ix
4 Exist
+ north → 2 four
2 He
+ 2 n
Neutrons are liberated when beryllium nuclei are struck by energetic alpha particles[8] producing the nuclear reaction
- 9
4 Be
+ 4
2 He
→ 12
6 C
+ n
where 4
two He
is an alpha particle and 12
6 C
is a carbon-12 nucleus.[9] Glucinium also releases neutrons under battery by gamma rays. Thus, natural beryllium bombarded either past alphas or gammas from a suitable radioisotope is a cardinal component of most radioisotope-powered nuclear reaction neutron sources for the laboratory production of free neutrons.
Small amounts of tritium are liberated when 9
4 Be
nuclei absorb low energy neutrons in the iii-footstep nuclear reaction
- nine
4 Be
+ n → 4
2 He
+ 6
two He
, 6
2 He
→ 6
3 Li
+ β−, half-dozen
3 Li
+ due north → 4
two He
+ three
1 H
Note that half-dozen
2 He
has a half-life of only 0.8 seconds, β− is an electron, and half dozen
three Li
has a high neutron absorption cross section. Tritium is a radioisotope of business organization in nuclear reactor waste streams.[10]
Optical backdrop [edit]
Equally a metal, glucinium is transparent or translucent to most wavelengths of X-rays and gamma rays, making it useful for the output windows of X-ray tubes and other such apparatus.[xi]
Isotopes and nucleosynthesis [edit]
Both stable and unstable isotopes of glucinium are created in stars, but the radioisotopes do not last long. Information technology is believed that nearly of the stable beryllium in the universe was originally created in the interstellar medium when cosmic rays induced fission in heavier elements constitute in interstellar gas and dust.[12] Primordial glucinium contains just 1 stable isotope, nineBe, and therefore beryllium is a monoisotopic and mononuclidic element.

Plot showing variations in solar activity, including variation in sunspot number (ruddy) and 10Be concentration (blue). Note that the beryllium calibration is inverted, so increases on this scale indicate lower 10Exist levels
Radioactive cosmogenic 10Be is produced in the atmosphere of the Earth by the cosmic ray spallation of oxygen.[thirteen] 10Be accumulates at the soil surface, where its relatively long half-life (i.36 million years) permits a long residence time before decaying to boron-10. Thus, 10Be and its daughter products are used to examine natural soil erosion, soil formation and the evolution of lateritic soils, and equally a proxy for measurement of the variations in solar action and the age of ice cores.[fourteen] The product of 10Exist is inversely proportional to solar activity, because increased solar current of air during periods of high solar activeness decreases the flux of galactic catholic rays that reach the Globe.[13] Nuclear explosions besides form 10Be by the reaction of fast neutrons with 13C in the carbon dioxide in air. This is one of the indicators of past activeness at nuclear weapon test sites.[15] The isotope sevenBe (half-life 53 days) is also cosmogenic, and shows an atmospheric abundance linked to sunspots, much similar 10Be.
viiiExist has a very short one-half-life of about 8×10 −17 s that contributes to its significant cosmological role, as elements heavier than glucinium could not have been produced by nuclear fusion in the Big Bang.[16] This is due to the lack of sufficient time during the Big Bang'south nucleosynthesis stage to produce carbon past the fusion of fourHe nuclei and the very low concentrations of available glucinium-8. British astronomer Sir Fred Hoyle commencement showed that the free energy levels of 8Exist and 12C allow carbon product by the so-chosen triple-alpha process in helium-fueled stars where more nucleosynthesis time is available. This process allows carbon to be produced in stars, but not in the Large Blindside. Star-created carbon (the ground of carbon-based life) is thus a component in the elements in the gas and dust ejected by AGB stars and supernovae (meet also Big Bang nucleosynthesis), besides as the creation of all other elements with diminutive numbers larger than that of carbon.[17]
The 2s electrons of glucinium may contribute to chemical bonding. Therefore, when 7Be decays by L-electron capture, it does so past taking electrons from its atomic orbitals that may be participating in bonding. This makes its decay charge per unit dependent to a measurable degree upon its chemical surroundings – a rare occurrence in nuclear decay.[xviii]
The shortest-lived known isotope of beryllium is 16Be, which decays through neutron emission with a half-life of half dozen.v×10−22 s.[19] The exotic isotopes elevenBe and 14Be are known to exhibit a nuclear halo.[20] This phenomenon can be understood equally the nuclei of xiBe and xivExist have, respectively, 1 and 4 neutrons orbiting substantially outside the classical Fermi 'waterdrop' model of the nucleus.
Occurrence [edit]

Beryllium ore with 1US¢ money for calibration

The Dominicus has a concentration of 0.ane parts per billion (ppb) of beryllium.[21] Beryllium has a concentration of 2 to 6 parts per million (ppm) in the Earth'southward crust.[22] It is most concentrated in the soils, 6 ppm.[23] Trace amounts of ixBe are institute in the Earth's atmosphere.[23] The concentration of beryllium in sea water is 0.ii–0.half-dozen parts per trillion.[23] [24] In stream water, however, beryllium is more abundant with a concentration of 0.1 ppb.[25]
Beryllium is found in over 100 minerals,[26] but about are uncommon to rare. The more than common beryllium containing minerals include: bertrandite (ExistivSiiiO7(OH)2), beryl (Al2Be3Sihalf dozenO18), chrysoberyl (Al2BeOfour) and phenakite (BeiiSiOiv). Precious forms of beryl are aquamarine, ruby beryl and emerald.[8] [27] [28] The green color in gem-quality forms of beryl comes from varying amounts of chromium (most two% for emerald).[29]
The 2 main ores of glucinium, beryl and bertrandite, are found in Argentine republic, Brazil, Republic of india, Republic of madagascar, Russian federation and the United States.[29] Full globe reserves of glucinium ore are greater than 400,000 tonnes.[29]
Production [edit]
The extraction of beryllium from its compounds is a hard process due to its high affinity for oxygen at elevated temperatures, and its ability to reduce h2o when its oxide pic is removed. Currently the United States, Mainland china and Kazakhstan are the just 3 countries involved in the industrial-scale extraction of glucinium.[30] Kazakhstan produces beryllium from a concentrate stockpiled before the breakdown of the Soviet Union around 1991. This resource has become nigh depleted past mid-2010s.[31]
Product of beryllium in Russia was halted in 1997, and is planned to be resumed in the 2020s.[32] [33]
Beryllium is most ordinarily extracted from the mineral beryl, which is either sintered using an extraction agent or melted into a soluble mixture. The sintering procedure involves mixing beryl with sodium fluorosilicate and soda at 770 °C (1,420 °F) to course sodium fluoroberyllate, aluminium oxide and silicon dioxide.[6] Beryllium hydroxide is precipitated from a solution of sodium fluoroberyllate and sodium hydroxide in water. Extraction of glucinium using the melt method involves grinding beryl into a powder and heating information technology to 1,650 °C (3,000 °F).[6] The melt is speedily cooled with h2o and and so reheated 250 to 300 °C (482 to 572 °F) in full-bodied sulfuric acid, by and large yielding beryllium sulfate and aluminium sulfate.[six] Aqueous ammonia is then used to remove the aluminium and sulfur, leaving beryllium hydroxide.
Beryllium hydroxide created using either the sinter or melt method is then converted into glucinium fluoride or beryllium chloride. To form the fluoride, aqueous ammonium hydrogen fluoride is added to beryllium hydroxide to yield a precipitate of ammonium tetrafluoroberyllate, which is heated to i,000 °C (1,830 °F) to form glucinium fluoride.[half dozen] Heating the fluoride to 900 °C (i,650 °F) with magnesium forms finely divided glucinium, and additional heating to one,300 °C (2,370 °F) creates the compact metal.[half dozen] Heating glucinium hydroxide forms the oxide, which becomes glucinium chloride when combined with carbon and chlorine. Electrolysis of molten glucinium chloride is then used to obtain the metal.[6]
Chemical backdrop [edit]
A beryllium cantlet has the electronic configuration [He] 2s2. The predominant oxidation state of beryllium is +2; the glucinium atom has lost both of its valence electrons. Lower oxidation states accept been establish in, for case, bis(carbene) compounds.[34] Beryllium's chemical behavior is largely a result of its modest atomic and ionic radii. Information technology thus has very high ionization potentials and potent polarization while bonded to other atoms, which is why all of its compounds are covalent. Its chemistry has similarities to that of aluminium, an case of a diagonal relationship.
At room temperature, the surface of beryllium forms a 1−10 nm-thick oxide passivation layer that prevents further reactions with air, except for gradual thickening of the oxide up to about 25 nm. When heated to a higher place about 500 °C, oxidation into the bulk metallic progresses forth grain boundaries.[35] Once the metal is ignited in air by heating to a higher place the oxide melting point effectually 2500 °C, beryllium burns brilliantly, forming a mixture of beryllium oxide and beryllium nitride. Beryllium dissolves readily in non-oxidizing acids, such as HCl and diluted HiiAnd soiv, simply not in nitric acid or water as this forms the oxide. This beliefs is like to that of aluminium metallic. Beryllium also dissolves in brine solutions.[6] [36]
Binary compounds of glucinium(II) are polymeric in the solid country. BeF2 has a silica-like structure with corner-shared BeFfour tetrahedra. BeCl2 and BeBr2 have chain structures with edge-shared tetrahedra. Glucinium oxide, BeO, is a white refractory solid, which has the wurtzite crystal structure and a thermal conductivity as high as some metals. BeO is amphoteric. Beryllium sulfide, selenide and telluride are known, all having the zincblende structure.[37] Glucinium nitride, Be3N2 is a loftier-melting-indicate compound which is readily hydrolyzed. Beryllium azide, BeN6 is known and beryllium phosphide, Be3P2 has a similar structure to BeiiiN2. A number of glucinium borides are known, such equally BefiveB, BefourB, Exist2B, BeB2, BeB6 and BeB12. Beryllium carbide, BeiiC, is a refractory brick-red compound that reacts with water to give methane.[37] No beryllium silicide has been identified.[36]
The halides BeX2 (10 = F, Cl, Br, I) have a linear monomeric molecular structure in the gas phase.[36] Complexes of the halides are formed with one or more ligands altruistic at total of two pairs of electrons. Such compounds obey the octet rule. Other 4-coordinate complexes such as the aqua-ion [Be(H2O)iv]ii+ also obey the octet rule.
Aqueous solutions [edit]
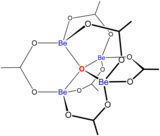
Schematic structure of bones beryllium acetate
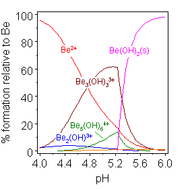
Beryllium hydrolysis. Water molecules attached to Be are omitted in this diagram
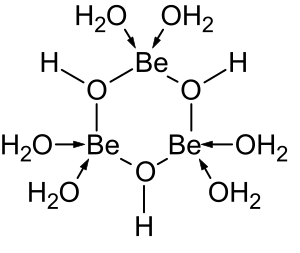
Structure of the trimeric hydrolysis product of beryllium(Two)
The aqueous solution chemistry of beryllium is the discipline of a comprehensive review.[38] Solutions of glucinium salts, such every bit beryllium sulfate and beryllium nitrate, are acidic considering of hydrolysis of the [Be(HiiO)4]2+ ion. The concentration of the showtime hydrolysis product, [Be(HiiO)3(OH)]+, is less than 1% of the beryllium concentration. The most stable hydrolysis production is the trimeric ion [Bethree(OH)three(H2O)6]three+. Glucinium hydroxide, Exist(OH)2, is insoluble in water at pH v or more. Consequently, beryllium compounds are generally insoluble at biological pH. Because of this, inhalation of beryllium metal dust past people leads to the development of the fatal status of berylliosis. Be(OH)two dissolves in strongly alkali metal solutions.
Beryllium(2) forms few complexes with monodentate ligands because the water molecules in the aquo-ion, are spring very strongly to the glucinium ion. Notable exceptions are the series of water-soluble complexes with the fluoride ion.[39]
Beryllium(2) forms many complexes with bidentate ligands containing oxygen-donor atoms.[38] The species is notable for having a 3-coordinate oxide ion at its center. Basic glucinium acetate, , has an oxide ion surrounded by a tetrahedron of beryllium atoms.
With organic ligands, such as the malonate ion, the acrid is de-protonated when forming the circuitous. The donor atoms are ii oxygens.
Formation of a complex is in competition with the metallic ion-hydrolysis reaction and mixed complexes with both the anion and the hydroxide ion are also formed. For example, derivatives of the cyclic trimer are known, with a bidentate ligand replacing ane or more pairs of water molecules. Ligands such every bit EDTA bear every bit dicarboxylic acids.
Hydroxycarboxylic acids such as glycollic acid form rather weak, monodentate, complexes in solution in which the hydroxyl grouping remains intact. A hexamer, , in which the hydroxyl groups are deprotonated was isolated, in the solid state, long agone.[40] Aromatic di-hydroxy ligands form relatively strong complexes. For example, log Kane and log Grandtwo values of 12.2 and ix.iii have been reported for complexes with tiron.[41]
There are many early reports of complexes with amino acids, but unfortunately they are not reliable every bit the concomitant hydrolysis reactions were not understood at the time of publication. Values for log β of ca. six to 7 have been reported.[42] The degree of formation is minor because of contest with hydrolysis reactions.
Organic chemistry [edit]
Organoberyllium chemical science is limited to bookish enquiry due to the cost and toxicity of beryllium, beryllium derivatives and reagents required for the introduction of beryllium, such as glucinium chloride. Organometallic glucinium compounds are known to exist highly reactive[43] Examples of known organoberyllium compounds are dineopentylberyllium,[44] beryllocene (Cp2Be),[45] [46] [47] [48] diallylberyllium (by substitution reaction of diethyl beryllium with triallyl boron),[49] bis(i,3-trimethylsilylallyl)beryllium[l] and Be(mes)2.[43] Ligands can as well be aryls[51] and alkynyls.[52]
History [edit]
The mineral beryl, which contains glucinium, has been used at least since the Ptolemaic dynasty of Egypt.[53] In the first century CE, Roman naturalist Pliny the Elderberry mentioned in his encyclopedia Natural History that beryl and emerald ("smaragdus") were similar.[54] The Papyrus Graecus Holmiensis, written in the third or fourth century CE, contains notes on how to set artificial emerald and beryl.[54]

Early analyses of emeralds and beryls by Martin Heinrich Klaproth, Torbern Olof Bergman, Franz Karl Achard, and Johann Jakob Bindheim always yielded similar elements, leading to the mistaken conclusion that both substances are aluminium silicates.[55] Mineralogist René Just Haüy discovered that both crystals are geometrically identical, and he asked pharmacist Louis-Nicolas Vauquelin for a chemic analysis.[53]
In a 1798 paper read earlier the Institut de French republic, Vauquelin reported that he found a new "earth" past dissolving aluminium hydroxide from emerald and beryl in an additional brine.[56] The editors of the journal Annales de Chimie et de Physique named the new earth "glucine" for the sugariness gustatory modality of some of its compounds.[57] Klaproth preferred the proper noun "beryllina" due to the fact that yttria likewise formed sweet salts.[58] [59] The name "beryllium" was get-go used by Wöhler in 1828.[60]

Friedrich Wöhler[61] and Antoine Bussy[62] independently isolated beryllium in 1828 by the chemical reaction of metallic potassium with beryllium chloride, as follows:
- BeClii + ii One thousand → ii KCl + Be
Using an alcohol lamp, Wöhler heated alternate layers of beryllium chloride and potassium in a wired-shut platinum crucible. The above reaction immediately took identify and acquired the crucible to become white hot. Upon cooling and washing the resulting gray-blackness powder he saw that it was fabricated of fine particles with a nighttime metal luster.[63] The highly reactive potassium had been produced past the electrolysis of its compounds, a process discovered 21 years before. The chemic method using potassium yielded only small grains of beryllium from which no ingot of metal could be bandage or hammered.
The direct electrolysis of a molten mixture of beryllium fluoride and sodium fluoride by Paul Lebeau in 1898 resulted in the start pure (99.5 to 99.eight%) samples of glucinium.[63] However, industrial product started just later the First Earth State of war. The original industrial interest included subsidiaries and scientists related to the Spousal relationship Carbide and Carbon Corporation in Cleveland, Ohio, and Siemens & Halske AG in Berlin. In the US, the process was ruled by Hugh Southward. Cooper, director of The Kemet Laboratories Visitor. In Federal republic of germany, the start commercially successful procedure for producing glucinium was developed in 1921 by Alfred Stock and Hans Goldschmidt.[64]
A sample of beryllium was bombarded with alpha rays from the decay of radium in a 1932 experiment by James Chadwick that uncovered the beingness of the neutron.[29] This same method is used in one grade of radioisotope-based laboratory neutron sources that produce 30 neutrons for every million α particles.[22]
Glucinium production saw a rapid increase during World War II, due to the ascent demand for hard beryllium-copper alloys and phosphors for fluorescent lights. Near early fluorescent lamps used zinc orthosilicate with varying content of beryllium to emit greenish light. Modest additions of magnesium tungstate improved the blue part of the spectrum to yield an acceptable white light. Halophosphate-based phosphors replaced beryllium-based phosphors afterwards beryllium was found to be toxic.[65]
Electrolysis of a mixture of glucinium fluoride and sodium fluoride was used to isolate beryllium during the 19th century. The metal's loftier melting point makes this process more than free energy-consuming than corresponding processes used for the alkali metals. Early in the 20th century, the production of beryllium by the thermal decomposition of beryllium iodide was investigated following the success of a similar process for the production of zirconium, just this process proved to be uneconomical for volume production.[66]
Pure beryllium metal did not go readily available until 1957, even though information technology had been used as an alloying metal to harden and toughen copper much earlier.[29] Beryllium could be produced past reducing beryllium compounds such as glucinium chloride with metallic potassium or sodium. Currently, most beryllium is produced by reducing beryllium fluoride with magnesium.[67] The price on the American market for vacuum-cast beryllium ingots was nearly $338 per pound ($745 per kilogram) in 2001.[68]
Betwixt 1998 and 2008, the earth's production of beryllium had decreased from 343 to about 200 tonnes. It then increased to 230 tonnes past 2018, of which 170 tonnes came from the United States.[69] [70]
Etymology [edit]
Named later beryl, a semiprecious mineral, from which it was first isolated.[71] [72] [73]
Applications [edit]
Radiation windows [edit]

Beryllium target which converts a proton beam into a neutron axle

A square beryllium foil mounted in a steel instance to be used equally a window between a vacuum chamber and an X-ray microscope. Beryllium is highly transparent to Ten-rays owing to its low atomic number.
Because of its low diminutive number and very low assimilation for Ten-rays, the oldest and still one of the well-nigh important applications of beryllium is in radiation windows for X-ray tubes.[29] Extreme demands are placed on purity and cleanliness of beryllium to avert artifacts in the 10-ray images. Thin beryllium foils are used as radiation windows for Ten-ray detectors, and the extremely low absorption minimizes the heating furnishings caused by high intensity, low energy X-rays typical of synchrotron radiation. Vacuum-tight windows and beam-tubes for radiations experiments on synchrotrons are manufactured exclusively from glucinium. In scientific setups for diverse 10-ray emission studies (e.grand., free energy-dispersive X-ray spectroscopy) the sample holder is unremarkably made of beryllium considering its emitted X-rays accept much lower energies (≈100 eV) than X-rays from most studied materials.[viii]
Depression atomic number besides makes beryllium relatively transparent to energetic particles. Therefore, information technology is used to build the beam pipage around the collision region in particle physics setups, such as all iv main detector experiments at the Large Hadron Collider (ALICE, ATLAS, CMS, LHCb),[74] the Tevatron and at SLAC. The low density of beryllium allows collision products to reach the surrounding detectors without significant interaction, its stiffness allows a powerful vacuum to be produced inside the pipage to minimize interaction with gases, its thermal stability allows it to office correctly at temperatures of only a few degrees above accented zero, and its diamagnetic nature keeps it from interfering with the circuitous multipole magnet systems used to steer and focus the particle beams.[75]
Mechanical applications [edit]
Because of its stiffness, light weight and dimensional stability over a broad temperature range, beryllium metal is used for lightweight structural components in the defense and aerospace industries in high-speed aircraft, guided missiles, spacecraft, and satellites, including the James Webb Space Telescope. Several liquid-fuel rockets accept used rocket nozzles fabricated of pure glucinium.[76] [77] Glucinium powder was itself studied as a rocket fuel, but this use has never materialized.[29] A small number of extreme high-end bicycle frames have been built with beryllium.[78] From 1998 to 2000, the McLaren Formula One team used Mercedes-Benz engines with glucinium-aluminium-alloy pistons.[79] The apply of beryllium engine components was banned following a protest by Scuderia Ferrari.[80]
Mixing well-nigh 2.0% glucinium into copper forms an alloy called beryllium copper that is six times stronger than copper alone.[81] Beryllium alloys are used in many applications because of their combination of elasticity, high electric conductivity and thermal conductivity, high strength and hardness, nonmagnetic properties, as well equally good corrosion and fatigue resistance.[29] [half dozen] These applications include non-sparking tools that are used near flammable gases (glucinium nickel), in springs and membranes (beryllium nickel and beryllium iron) used in surgical instruments and loftier temperature devices.[29] [half dozen] As little as 50 parts per million of beryllium alloyed with liquid magnesium leads to a significant increase in oxidation resistance and decrease in flammability.[vi]

Beryllium copper adaptable wrench
The loftier elastic stiffness of beryllium has led to its all-encompassing use in precision instrumentation, e.chiliad. in inertial guidance systems and in the support mechanisms for optical systems.[8] Beryllium-copper alloys were likewise applied as a hardening agent in "Jason pistols", which were used to strip the paint from the hulls of ships.[82]
Beryllium was too used for cantilevers in loftier performance phonograph cartridge styli, where its farthermost stiffness and depression density immune for tracking weights to be reduced to 1 gram, yet still track high frequency passages with minimal baloney.[83]
An earlier major application of beryllium was in brakes for military machine airplanes considering of its hardness, loftier melting point, and exceptional ability to dissipate oestrus. Environmental considerations have led to substitution by other materials.[eight]
To reduce costs, beryllium can be alloyed with significant amounts of aluminium, resulting in the AlBeMet alloy (a trade name). This blend is cheaper than pure beryllium, while nonetheless retaining many desirable backdrop.
Mirrors [edit]
Beryllium mirrors are of particular interest. Large-area mirrors, frequently with a honeycomb support structure, are used, for instance, in meteorological satellites where low weight and long-term dimensional stability are critical. Smaller glucinium mirrors are used in optical guidance systems and in burn down-control systems, east.thousand. in the German language-fabricated Leopard i and Leopard two master battle tanks. In these systems, very rapid movement of the mirror is required which over again dictates low mass and high rigidity. Usually the beryllium mirror is coated with hard electroless nickel plating which tin be more than hands polished to a finer optical stop than beryllium. In some applications, though, the beryllium blank is polished without any coating. This is particularly applicable to cryogenic operation where thermal expansion mismatch tin crusade the blanket to buckle.[8]
The James Webb Space Telescope has xviii hexagonal beryllium sections for its mirrors, each plated with a thin layer of gold.[84] Considering JWST will confront a temperature of 33 K, the mirror is made of aureate-plated beryllium, capable of handling extreme cold meliorate than glass. Beryllium contracts and deforms less than glass – and remains more than uniform – in such temperatures.[85] For the same reason, the eyes of the Spitzer Space Telescope are entirely built of beryllium metal.[86]
Magnetic applications [edit]
Beryllium is not-magnetic. Therefore, tools fabricated out of beryllium-based materials are used by naval or military explosive ordnance disposal teams for work on or virtually naval mines, since these mines unremarkably have magnetic fuzes.[88] They are also found in maintenance and construction materials near magnetic resonance imaging (MRI) machines because of the high magnetic fields generated.[89] In the fields of radio communications and powerful (usually military) radars, manus tools fabricated of beryllium are used to tune the highly magnetic klystrons, magnetrons, traveling wave tubes, etc., that are used for generating loftier levels of microwave power in the transmitters.[90]
Nuclear applications [edit]
Thin plates or foils of beryllium are sometimes used in nuclear weapon designs every bit the very outer layer of the plutonium pits in the master stages of thermonuclear bombs, placed to surround the fissile material. These layers of beryllium are good "pushers" for the implosion of the plutonium-239, and they are good neutron reflectors, just as in beryllium-chastened nuclear reactors.[91]
Beryllium is likewise commonly used in some neutron sources in laboratory devices in which relatively few neutrons are needed (rather than having to apply a nuclear reactor, or a particle accelerator-powered neutron generator). For this purpose, a target of glucinium-9 is bombarded with energetic alpha particles from a radioisotope such as polonium-210, radium-226, plutonium-238, or americium-241. In the nuclear reaction that occurs, a beryllium nucleus is transmuted into carbon-12, and i free neutron is emitted, traveling in about the same management every bit the alpha particle was heading. Such blastoff disuse driven beryllium neutron sources, named "urchin" neutron initiators, were used in some early atomic bombs.[91] Neutron sources in which beryllium is bombarded with gamma rays from a gamma decay radioisotope, are also used to produce laboratory neutrons.[92]

Two CANDU fuel bundles: Each about 50 cm in length and 10 cm in diameter. Detect the small appendages on the fuel clad surfaces
Beryllium is also used in fuel fabrication for CANDU reactors. The fuel elements have small appendages that are resistance brazed to the fuel cladding using an induction brazing process with Be as the braze filler material. Bearing pads are brazed in place to forestall contact between the fuel bundle and the pressure level tube containing it, and inter-chemical element spacer pads are brazed on to foreclose element to element contact.
Glucinium is also used at the Joint European Torus nuclear-fusion research laboratory, and it volition be used in the more advanced ITER to condition the components which confront the plasma.[93] Glucinium has also been proposed as a cladding material for nuclear fuel rods, considering of its skillful combination of mechanical, chemical, and nuclear properties.[8] Beryllium fluoride is one of the constituent salts of the eutectic salt mixture FLiBe, which is used as a solvent, moderator and coolant in many hypothetical molten salt reactor designs, including the liquid fluoride thorium reactor (LFTR).[94]
Acoustics [edit]
The low weight and loftier rigidity of beryllium make it useful as a textile for high-frequency speaker drivers. Because beryllium is expensive (many times more than titanium), difficult to shape due to its brittleness, and toxic if mishandled, beryllium tweeters are express to loftier-end dwelling,[95] [96] [97] pro audio, and public address applications.[98] [99] Some high-fidelity products have been fraudulently claimed to be fabricated of the cloth.[100]
Some loftier-stop phonograph cartridges used glucinium cantilevers to improve tracking past reducing mass.[101]
Electronic [edit]
Beryllium is a p-blazon dopant in 3-V compound semiconductors. It is widely used in materials such every bit GaAs, AlGaAs, InGaAs and InAlAs grown by molecular beam epitaxy (MBE).[102] Cross-rolled beryllium sheet is an splendid structural back up for printed circuit boards in surface-mount engineering science. In critical electronic applications, beryllium is both a structural support and heat sink. The awarding as well requires a coefficient of thermal expansion that is well matched to the alumina and polyimide-glass substrates. The beryllium-beryllium oxide composite "E-Materials" accept been specially designed for these electronic applications and have the boosted reward that the thermal expansion coefficient can exist tailored to match diverse substrate materials.[8]
Glucinium oxide is useful for many applications that crave the combined properties of an electrical insulator and an excellent heat usher, with high strength and hardness, and a very high melting point. Beryllium oxide is oftentimes used every bit an insulator base plate in high-power transistors in radio frequency transmitters for telecommunications. Beryllium oxide is too existence studied for use in increasing the thermal conductivity of uranium dioxide nuclear fuel pellets.[103] Beryllium compounds were used in fluorescent lighting tubes, just this use was discontinued considering of the affliction berylliosis which developed in the workers who were making the tubes.[104]
Healthcare [edit]
Glucinium is a component of several dental alloys.[105] [106]
Toxicity and safety [edit]
| Hazards | |
|---|---|
| GHS labelling:[107] | |
| Pictograms |   |
| Signal word | Danger |
| Hazard statements | H301, H315, H317, H319, H330, H335, H350i, H372 |
| Precautionary statements | P201, P202, P280, P302, P304, P305+P351+P338, P310, P340, P352 |
| NFPA 704 (fire diamond) | 4 three iii |
Biological effects [edit]
Approximately 35 micrograms of beryllium is found in the boilerplate homo body, an corporeality not considered harmful.[108] Glucinium is chemically similar to magnesium and therefore can displace it from enzymes, which causes them to malfunction.[108] Because Be2+ is a highly charged and small ion, it can easily get into many tissues and cells, where it specifically targets cell nuclei, inhibiting many enzymes, including those used for synthesizing DNA. Its toxicity is exacerbated by the fact that the body has no means to control beryllium levels, and once inside the torso the beryllium cannot exist removed.[109]
Inhalation [edit]
Chronic berylliosis is a pulmonary and systemic granulomatous disease acquired by inhalation of dust or fumes contaminated with beryllium; either big amounts over a short time or modest amounts over a long fourth dimension can lead to this ailment. Symptoms of the disease tin can take up to v years to develop; about a third of patients with it die and the survivors are left disabled.[108] The International Agency for Research on Cancer (IARC) lists beryllium and glucinium compounds as Category one carcinogens.[110]
Occupational exposure [edit]
In the US, the Occupational Safety and Wellness Administration (OSHA) has designated a permissible exposure limit (PEL) in the workplace with a time-weighted boilerplate (TWA) 2 µg/m3 and a constant exposure limit of 5 µg/mthree over 30 minutes, with a maximum height limit of 25 µg/giii. The National Constitute for Occupational Safety and Health (NIOSH) has ready a recommended exposure limit (REL) of constant 500 ng/chiliad3. The IDLH (immediately dangerous to life and health) value is 4 mg/yard3.[111] The toxicity of glucinium is on par with other toxic metals, such as arsenic and mercury.[112] [113]
Exposure to beryllium in the workplace can atomic number 82 to a sensitization immune response and can over time develop chronic beryllium disease (CBD).[114] The National Institute for Occupational Safety and Health (NIOSH) in the Us researches these furnishings in collaboration with a major manufacturer of beryllium products. NIOSH also conducts genetic enquiry on sensitization and CBD, independently of this collaboration.[114]
Acute glucinium affliction in the form of chemic pneumonitis was first reported in Europe in 1933 and in the Usa in 1943. A survey constitute that about 5% of workers in plants manufacturing fluorescent lamps in 1949 in the United States had beryllium-related lung diseases.[115] Chronic berylliosis resembles sarcoidosis in many respects, and the differential diagnosis is oftentimes difficult. Information technology killed some early workers in nuclear weapons design, such every bit Herbert L. Anderson.[116]
Beryllium may exist found in coal slag. When the slag is formulated into an abrasive amanuensis for blasting paint and rust from hard surfaces, the beryllium can become airborne and become a source of exposure.[117]
Although the use of glucinium compounds in fluorescent lighting tubes was discontinued in 1949, potential for exposure to glucinium exists in the nuclear and aerospace industries and in the refining of beryllium metal and melting of beryllium-containing alloys, the manufacturing of electronic devices, and the handling of other glucinium-containing material.[118]
Detection [edit]
Early researchers undertook the highly hazardous practice of identifying beryllium and its various compounds from its sweetness gustation. Identification is now performed using rubber modernistic diagnostics techniques.[six] A successful test for glucinium in air and on surfaces has been developed and published as an international voluntary consensus standard ASTM D7202. The process uses dilute ammonium bifluoride for dissolution and fluorescence detection with beryllium bound to sulfonated hydroxybenzoquinoline, allowing up to 100 times more than sensitive detection than the recommended limit for glucinium concentration in the workplace. Fluorescence increases with increasing beryllium concentration. The new process has been successfully tested on a variety of surfaces and is effective for the dissolution and detection of refractory beryllium oxide and siliceous glucinium in infinitesimal concentrations (ASTM D7458).[119] [120] The NIOSH Manual of Analytical Methods contains methods for measuring occupational exposures to beryllium.[121]
References [edit]
- ^ "Standard Atomic Weights: Beryllium". CIAAW. 2013.
- ^ Be(0) has been observed; meet "Beryllium(0) Complex Institute". Chemistry Europe. 13 June 2016.
- ^ "Beryllium: Beryllium(I) Hydride chemical compound information" (PDF). bernath.uwaterloo.ca. Retrieved ten December 2007.
- ^ Haynes, William M., ed. (2011). CRC Handbook of Chemistry and Physics (92nd ed.). Boca Raton, FL: CRC Press. p. 14.48. ISBN1-4398-5511-0.
- ^ Weast, Robert (1984). CRC, Handbook of Chemistry and Physics. Boca Raton, Florida: Chemical Rubber Company Publishing. pp. E110. ISBN0-8493-0464-4.
- ^ a b c d east f g h i j k 50 grand northward o Jakubke, Hans-Dieter; Jeschkeit, Hans, eds. (1994). Concise Encyclopedia Chemical science. trans. rev. Eagleson, Mary. Berlin: Walter de Gruyter.
- ^ Puchta, Ralph (2011). "A brighter beryllium". Nature Chemistry. three (five): 416. Bibcode:2011NatCh...three..416P. doi:10.1038/nchem.1033. PMID 21505503.
- ^ a b c d e f thousand h i Behrens, V. (2003). "11 Beryllium". In Beiss, P. (ed.). Landolt-Börnstein – Group 8 Avant-garde Materials and Technologies: Powder Metallurgy Data. Refractory, Hard and Intermetallic Materials. Landolt-Börnstein - Group VIII Advanced Materials and Technologies. Vol. 2A1. Berlin: Springer. pp. 667–677. doi:ten.1007/10689123_36. ISBN978-3-540-42942-5.
- ^ a b Hausner, Henry H. (1965). "Nuclear Backdrop". Glucinium its Metallurgy and Properties. Academy of California Printing. p. 239. Archived from the original on 27 July 2020. Retrieved 30 October 2021.
- ^ Tomberlin, T. A. (15 November 2004). "Beryllium – A Unique Fabric in Nuclear Applications" (PDF). Idaho National Laboratory. Idaho National Engineering and Environmental Laboratory. Archived from the original (PDF) on 22 December 2015.
- ^ "About Beryllium". Us Section of Free energy. Archived from the original on 22 Dec 2021. Retrieved 22 December 2021.
- ^ Ekspong, G. (1992). Physics: 1981–1990. World Scientific. pp. 172 ff. ISBN978-981-02-0729-8. Archived from the original on 27 July 2020. Retrieved 30 Oct 2021.
- ^ a b Emsley 2001, p. 56.
- ^ "Beryllium: Isotopes and Hydrology". University of Arizona, Tucson. Archived from the original on 26 May 2013. Retrieved x April 2011.
- ^ Whitehead, N; Endo, S; Tanaka, Yard; Takatsuji, T; Hoshi, M; Fukutani, S; Ditchburn, Rg; Zondervan, A (Feb 2008). "A preliminary study on the use of (10)Be in forensic radioecology of nuclear explosion sites". Journal of Environmental Radioactivity. 99 (2): 260–seventy. doi:10.1016/j.jenvrad.2007.07.016. PMID 17904707.
- ^ Boyd, R. N.; Kajino, T. (1989). "Tin Be-ix provide a exam of cosmological theories?". The Astrophysical Journal. 336: L55. Bibcode:1989ApJ...336L..55B. doi:10.1086/185360.
- ^ Arnett, David (1996). Supernovae and nucleosynthesis. Princeton University Press. p. 223. ISBN978-0-691-01147-9. Archived from the original on 27 July 2020. Retrieved thirty October 2021.
- ^ Johnson, Bill (1993). "How to Change Nuclear Decay Rates". University of California, Riverside. Archived from the original on 29 June 2013. Retrieved xxx March 2008.
- ^ Hammond, C. R. "Elements" in Lide, D. R., ed. (2005). CRC Handbook of Chemical science and Physics (86th ed.). Boca Raton (FL): CRC Press. ISBN0-8493-0486-5.
- ^ Hansen, P. K.; Jensen, A. S.; Jonson, B. (1995). "Nuclear Halos". Annual Review of Nuclear and Particle Science. 45 (45): 591–634. Bibcode:1995ARNPS..45..591H. doi:10.1146/annurev.ns.45.120195.003111.
- ^ "Abundance in the dominicus". Marker Winter, The University of Sheffield and WebElements Ltd, Britain. WebElements. Archived from the original on 27 Baronial 2011. Retrieved half-dozen August 2011.
- ^ a b O'Neil, Marydale J.; Heckelman, Patricia Eastward.; Roman, Cherie B., eds. (2006). The Merck Alphabetize: An Encyclopedia of Chemicals, Drugs, and Biologicals (14th ed.). Whitehouse Station, NJ, United states of america: Merck Research Laboratories, Merck & Co., Inc. ISBN978-0-911910-00-ane.
- ^ a b c Emsley 2001, p. 59.
- ^ "Abundance in oceans". Marking Wintertime, The University of Sheffield and WebElements Ltd, United kingdom. WebElements. Archived from the original on 5 August 2011. Retrieved 6 Baronial 2011.
- ^ "Affluence in stream water". Mark Winter, The University of Sheffield and WebElements Ltd, UK. WebElements. Archived from the original on 4 August 2011. Retrieved 6 August 2011.
- ^ "Search Minerals By Chemical science". world wide web.mindat.org. Archived from the original on vi August 2021. Retrieved xxx October 2021.
- ^ Walsh, Kenneth A (2009). "Sources of Beryllium". Beryllium chemistry and processing. pp. 20–26. ISBN978-0-87170-721-v. Archived from the original on xiii May 2016. Retrieved 5 January 2016.
- ^ Mining, Society for Metallurgy, Exploration (U.S) (5 March 2006). "Distribution of major deposits". Industrial minerals & rocks: commodities, markets, and uses. pp. 265–269. ISBN978-0-87335-233-8. Archived from the original on 4 May 2016. Retrieved v January 2016.
{{cite book}}: CS1 maint: uses authors parameter (link) - ^ a b c d eastward f g h i Emsley 2001, p. 58.
- ^ "Sources of Beryllium". Materion Corporation. Archived from the original on 24 Dec 2016. Retrieved 23 December 2016.
- ^ "Beryllim" Archived 3 July 2021 at the Wayback Machine in 2016 Minerals Yearbook. USGS (September 2018).
- ^ Уральский производитель изумрудов планирует выпускать стратегический металл бериллий Archived 11 October 2021 at the Wayback Car. TASS.ru (15 May 2019)
- ^ "Russian federation restarts beryllium product after 20 years". Eurasian Business Briefing. xx Feb 2015. Archived from the original on 31 July 2017. Retrieved 22 February 2018.
- ^ Arrowsmith, Merle; Braunschweig, Holger; Celik, Mehmet Ali; Dellermann, Theresa; Dewhurst, Rian D.; Ewing, William C.; Hammond, Kai; Kramer, Thomas; Krummenacher, Ivo (2016). "Neutral zero-valent south-cake complexes with stiff multiple bonding". Nature Chemistry. 8 (9): 890–894. Bibcode:2016NatCh...viii..890A. doi:x.1038/nchem.2542. PMID 27334631.
- ^ Tomastik, C.; Werner, W.; Stori, H. (2005). "Oxidation of beryllium—a scanning Auger investigation". Nucl. Fusion. 45 (9): 1061. Bibcode:2005NucFu..45.1061T. doi:x.1088/0029-5515/45/9/005. S2CID 111381179.
- ^ a b c Greenwood, Norman Due north.; Earnshaw, Alan (1997). Chemical science of the Elements (2nd ed.). Butterworth-Heinemann. ISBN978-0-08-037941-8.
- ^ a b Wiberg, Egon; Holleman, Arnold Frederick (2001). Inorganic Chemistry. Elsevier. ISBN978-0-12-352651-9.
- ^ a b Alderghi, Lucia; Gans, Peter; Midollini, Stefano; Vacca, Alberto (2000). Sykes, A.G; Cowley, Alan, H. (eds.). "Aqueous Solution Chemistry of Glucinium". Advances in Inorganic Chemistry. San Diego: Academic Press. 50: 109–172. doi:10.1016/S0898-8838(00)50003-eight. ISBN9780120236503.
- ^ Bong, Northward.A. (1972). Advances in Inorganic Chemical science and Radiochemistry. Vol. xiv. New York: Academic Printing. pp. 256–277. doi:10.1016/S0065-2792(08)60008-4. ISBN978-0-12-023614-5.
- ^ Rosenheim, Arthur; Lehmann, Fritz (1924). "Über innerkomplexe Beryllate". Liebigs Ann. Chem. 440: 153–166. doi:10.1002/jlac.19244400115.
- ^ Schmidt, 1000.; Bauer, A.; Schier, A.; Schmidtbauer, H (1997). "Glucinium Chelation by Dicarboxylic Acids in Aqueous Solution". Z. Naturforsch. 53b (10): 2040–2043. doi:10.1021/ic961410k. PMID 11669821.
- ^ Mederos, A.; Dominguez, Southward.; Chinea, E.; Brito, F.; Middolini, S.; Vacca, A. (1997). "Recent aspects of the coordination chemical science of the very toxic cation beryllium(2): The search for sequestering agents". Bol. Soc. Chil. Quim. 42: 281.
- ^ a b Naglav, D.; Buchner, M. R.; Bendt, G.; Kraus, F. and Schulz, S. (2016). "Off the Browbeaten Track—A Hitchhiker'southward Guide to Glucinium Chemistry". Angew. Chem. Int. Ed. 55 (36): 10562–10576. doi:10.1002/anie.201601809. PMID 27364901.
{{cite periodical}}: CS1 maint: multiple names: authors list (link) - ^ Coates, One thousand. Eastward.; Francis, B. R. (1971). "Grooming of base-complimentary beryllium alkyls from trialkylboranes. Dineopentylberyllium, bis((trimethylsilyl)methyl)glucinium, and an ethylberyllium hydride". Journal of the Chemical Society A: Inorganic, Concrete, Theoretical: 1308. doi:10.1039/J19710001308.
- ^ Fischer, Ernst Otto; Hofmann, Hermann P. (1959). "Über Aromatenkomplexe von Metallen, XXV. Di-cyclopentadienyl-beryllium". Chemische Berichte. 92 (two): 482. doi:10.1002/cber.19590920233.
- ^ Nugent, Chiliad. Due west.; Beattie, J. Thou.; Hambley, T. West.; Snow, M. R. (1984). "A precise low-temperature crystal structure of Bis(cyclopentadienyl)glucinium". Australian Journal of Chemistry. 37 (8): 1601. doi:10.1071/CH9841601. S2CID 94408686.
- ^ Almenningen, A.; Haaland, Arne; Lusztyk, Janusz (1979). "The molecular construction of beryllocene, (C5H5)2Be. A reinvestigation past gas phase electron diffraction". Journal of Organometallic Chemistry. 170 (3): 271. doi:10.1016/S0022-328X(00)92065-5.
- ^ Wong, C. H.; Lee, T. Y.; Chao, K. J.; Lee, S. (1972). "Crystal construction of bis(cyclopentadienyl)beryllium at −120 °C". Acta Crystallographica Section B. 28 (6): 1662. doi:x.1107/S0567740872004820.
- ^ Wiegand, G.; Thiele, K.-H. (1974). "Ein Beitrag zur Existenz von Allylberyllium- und Allylaluminiumverbindungen". Zeitschrift für Anorganische und Allgemeine Chemie (in German). 405: 101–108. doi:10.1002/zaac.19744050111.
- ^ Chmely, Stephen C.; Hanusa, Timothy P.; Brennessel, William W. (2010). "Bis(one,iii-trimethylsilylallyl)beryllium". Angewandte Chemie International Edition. 49 (34): 5870–5874. doi:10.1002/anie.201001866. PMID 20575128.
- ^ Ruhlandt-Senge, Karin; Bartlett, Ruth A.; Olmstead, Marilyn M.; Power, Philip P. (1993). "Synthesis and structural characterization of the beryllium compounds [Exist(two,4,6-Me3C6H2)ii(OEt2)], [Be{O(two,4,6-tert-Bu3C6H2)}2(OEtii)], and [Be{South(2,4,6-tert-Bu3C6H2)}2(THF)]⋅PhMe and determination of the structure of [BeCl2(OEttwo)two]". Inorganic Chemistry. 32 (9): 1724–1728. doi:10.1021/ic00061a031.
- ^ Morosin, B.; Howatson, J. (1971). "The crystal construction of dimeric methyl-ane-propynyl- beryllium-trimethylamine". Journal of Organometallic Chemistry. 29: 7–xiv. doi:10.1016/S0022-328X(00)87485-9.
- ^ a b Weeks 1968, p. 535.
- ^ a b Weeks 1968, p. 536.
- ^ Weeks 1968, p. 537.
- ^ Vauquelin, Louis-Nicolas (1798). "De l'Aiguemarine, ou Béril; et découverie d'une terre nouvelle dans cette pierre" [Aquamarine or beryl; and discovery of a new earth in this rock]. Annales de Chimie. 26: 155–169. Archived from the original on 27 April 2016. Retrieved 5 January 2016.
- ^ In a footnote on page 169 Archived 23 June 2016 at the Wayback Machine of (Vauquelin, 1798), the editors write: "(1) La propriété la plus caractéristique de cette terre, confirmée par les dernières expériences de notre collègue, étant de former des sels d'une saveur sucrée, nous proposons de l'appeler glucine, de γλυκυς, doux, γλυκύ, vin doux, γλυκαιτω, rendre doux … Note des Rédacteurs." ((1) The most characteristic property of this world, confirmed past the recent experiments of our colleague [Vauquelin], being to form salts with a sugariness taste, we advise to call it glucine from γλυκυς, sweet, γλυκύ, sugariness wine, γλυκαιτω, to make sweet … Notation of the editors.)
- ^ Klaproth, Martin Heinrich, Beitrage zur Chemischen Kenntniss der Mineralkörper (Contribution to the chemical knowledge of mineral substances), vol. 3, (Berlin, (Frg): Heinrich August Rottmann, 1802), pages 78–79 Archived 26 April 2016 at the Wayback Car: "Als Vauquelin der von ihm im Beryll und Smaragd entdeckten neuen Erde, wegen ihrer Eigenschaft, süsse Mittelsalze zu bilden, den Namen Glykine, Süsserde, beilegte, erwartete er wohl nicht, dass sich bald nachher eine anderweitige Erde finden würde, welche mit völlig gleichem Rechte Anspruch an diesen Namen machen können. Um daher keine Verwechselung derselben mit der Yttererde zu veranlassen, würde es vielleicht gerathen seyn, jenen Namen Glykine aufzugeben, und durch Beryllerde (Beryllina) zu ersetzen; welche Namensveränderung auch bereits vom Hrn. Prof. Link, und zwar aus dem Grunde empfohlen worden, weil schon ein Pflanzengeschlecht Glycine vorhanden ist." (When Vauquelin conferred – on account of its property of forming sweet salts – the name glycine, sweet-globe, on the new globe that had been establish past him in beryl and smaragd, he certainly didn't expect that soon thereafter another globe would be found which with fully equal right could claim this name. Therefore, in gild to avoid defoliation of it with yttria-earth, it would possibly exist advisable to carelessness this name glycine and replace information technology with beryl-earth (beryllina); which name change was too recommended past Prof. Link, and for the reason that a genus of plants, Glycine, already exists.)
- ^ Weeks 1968, p. 538.
- ^ Wöhler, F. (1828). "Ueber das Beryllium und Yttrium" [On beryllium and yttrium]. Annalen der Physik und Chemie. 13 (89): 577–582. Bibcode:1828AnP....89..577W. doi:x.1002/andp.18280890805. Archived from the original on 26 April 2016. Retrieved 5 Jan 2016.
- ^ Wöhler, Friedrich (1828). "Ueber das Beryllium und Yttrium". Annalen der Physik und Chemie. 89 (8): 577–582. Bibcode:1828AnP....89..577W. doi:10.1002/andp.18280890805. Archived from the original on 27 May 2016. Retrieved 5 January 2016.
- ^ Bussy, Antoine (1828). "D'une travail qu'il a entrepris sur le glucinium". Periodical de Chimie Médicale (4): 456–457. Archived from the original on 22 May 2016. Retrieved 5 January 2016.
- ^ a b Weeks 1968, p. 539.
- ^ Boillat, Johann (27 Baronial 2016). From Raw Fabric to Strategic Alloys. The Instance of the International Beryllium Industry (1919–1939). 1st Globe Congress on Business History, At Bergen – Norway. doi:10.13140/rg.2.ii.35545.11363. Archived from the original on 30 Oct 2021. Retrieved xxx Oct 2021.
- ^ Kane, Raymond; Sell, Heinz (2001). "A Review of Early Inorganic Phosphors". Revolution in lamps: a chronicle of fifty years of progress. p. 98. ISBN978-0-88173-378-5. Archived from the original on 7 May 2016. Retrieved 5 Jan 2016.
- ^ Babu, R. Due south.; Gupta, C. G. (1988). "Beryllium Extraction – A Review". Mineral Processing and Extractive Metallurgy Review. four: 39–94. doi:10.1080/08827508808952633.
- ^ Hammond, C.R. (2003). "The Elements". CRC handbook of chemistry and physics (84th ed.). Boca Raton, FL: CRC Printing. pp. 4–5. ISBN978-0-8493-0595-v. Archived from the original on xiii March 2020. Retrieved eighteen July 2019.
- ^ "Glucinium Statistics and Information". United States Geological Survey. Archived from the original on sixteen September 2008. Retrieved xviii September 2008.
- ^ "Commodity Summary: Glucinium" (PDF). Usa Geological Survey. Archived (PDF) from the original on i June 2010. Retrieved 16 May 2010.
- ^ "Commodity Summary 2000: Beryllium" (PDF). United States Geological Survey. Archived (PDF) from the original on 16 July 2010. Retrieved 16 May 2010.
- ^ "etymology online". Archived from the original on 30 Oct 2020. Retrieved xxx October 2021.
- ^ "Encyclopædia Britannica". Archived from the original on 23 October 2021. Retrieved 30 October 2021.
- ^ "Elemental Matter". Archived from the original on 29 November 2020. Retrieved 30 Oct 2021.
- ^ Veness, R.; Ramos, D.; Lepeule, P.; Rossi, A.; Schneider, 1000.; Blanchard, S. "Installation and commissioning of vacuum systems for the LHC particle detectors" (PDF). CERN. Archived (PDF) from the original on 14 November 2011. Retrieved 13 Jan 2012.
- ^ Wieman, H; Bieser, F.; Kleinfelder, Due south.; Matis, H. S.; Nevski, P.; Rai, 1000.; Smirnov, North. (2001). "A new inner vertex detector for STAR" (PDF). Nuclear Instruments and Methods in Physics Research Section A. 473 (1–2): 205. Bibcode:2001NIMPA.473..205W. doi:10.1016/S0168-9002(01)01149-4. S2CID 39909027. Archived (PDF) from the original on 17 Oct 2020. Retrieved 30 October 2021.
- ^ Davis, Joseph R. (1998). "Glucinium". Metals handbook. ASM International. pp. 690–691. ISBN978-0-87170-654-vi. Archived from the original on 27 July 2020. Retrieved 30 Oct 2021.
- ^ Schwartz, Mel M. (2002). Encyclopedia of materials, parts, and finishes. CRC Press. p. 62. ISBN978-i-56676-661-6. Archived from the original on 27 July 2020. Retrieved xxx Oct 2021.
- ^ "Museum of Mountain Wheel Art & Technology: American Bicycle Manufacturing". Archived from the original on 20 July 2011. Retrieved 26 September 2011.
- ^ Ward, Wayne. "Aluminium-Beryllium". Ret-Monitor. Archived from the original on one August 2010. Retrieved 18 July 2012.
- ^ Collantine, Keith (8 February 2007). "Banned! – Beryllium". Archived from the original on 21 July 2012. Retrieved 18 July 2012.
- ^ Geller, Elizabeth, ed. (2004). Concise Encyclopedia of Chemical science. New York Metropolis: McGraw-Hill. ISBN978-0-07-143953-4.
- ^ "Defence forces face rare toxic metal exposure risk". The Sydney Morn Herald. one February 2005. Archived from the original on 30 Dec 2007. Retrieved 8 August 2009.
- ^ Shure V15VxMR user's guide, Page 2
- ^ "The Webb Space Telescope Will Rewrite Cosmic History. If It Works". Quanta Magazine. 3 December 2021. Archived from the original on 5 December 2021. Retrieved five December 2021.
- ^ Gardner, Jonathan P. (2007). "The James Webb Space Telescope" (PDF). Proceedings of Science: v. Bibcode:2007mru..confE...5G. Archived (PDF) from the original on 4 June 2016. Retrieved xv January 2009.
- ^ Werner, 1000. Westward.; Roellig, T. Fifty.; Low, F. J.; Rieke, One thousand. H.; Rieke, One thousand.; Hoffmann, W. F.; Young, E.; Houck, J. R.; et al. (2004). "The Spitzer Space Telescope Mission". Astrophysical Journal Supplement. 154 (ane): 1–9. arXiv:astro-ph/0406223. Bibcode:2004ApJS..154....1W. doi:10.1086/422992. S2CID 119379934.
- ^ Greyness, Theodore. Gyroscope sphere. An example of the chemical element Beryllium Archived 14 April 2021 at the Wayback Machine. periodictable.com
- ^ Kojola, Kenneth; Lurie, William (9 August 1961). "The choice of low-magnetic alloys for EOD tools". Naval Weapons Plant Washington DC. Archived from the original on 23 Baronial 2011. Retrieved 28 February 2010.
- ^ Dorsch, Jerry A. & Dorsch, Susan E. (2007). Agreement anesthesia equipment. Lippincott Williams & Wilkins. p. 891. ISBN978-0-7817-7603-five. Archived from the original on 27 July 2020. Retrieved 30 Oct 2021.
- ^ Ropp, Richard C. (31 December 2012). Encyclopedia of the Alkaline World Compounds. p. 7. ISBN9780444595539. Archived from the original on 11 May 2016. Retrieved 5 January 2016.
- ^ a b Barnaby, Frank (1993). How nuclear weapons spread. Routledge. p. 35. ISBN978-0-415-07674-six. Archived from the original on 27 July 2020. Retrieved thirty October 2021.
- ^ Byrne, J. Neutrons, Nuclei, and Thing, Dover Publications, Mineola, NY, 2011, ISBN 0486482383, pp. 32–33.
- ^ Clark, R. E. H.; Reiter, D. (2005). Nuclear fusion research. Springer. p. 15. ISBN978-three-540-23038-0. Archived from the original on 27 July 2020. Retrieved 30 October 2021.
- ^ Petti, D.; Smolik, G.; Simpson, M.; Sharpe, J.; Anderl, R.; Fukada, S.; Hatano, Y.; Hara, M.; et al. (2006). "JUPITER-II molten salt Flibe research: An update on tritium, mobilization and redox chemistry experiments". Fusion Engineering and Blueprint. 81 (eight–14): 1439. doi:10.1016/j.fusengdes.2005.08.101. OSTI 911741. Archived from the original on 26 Apr 2021. Retrieved xxx Oct 2021.
- ^ "Scan Speak offers Be tweeters to OEMs and Do-Information technology-Yourselfers" (PDF). Scan Speak. May 2010. Archived from the original (PDF) on 3 March 2016.
- ^ Johnson, John Eastward. Jr. (12 November 2007). "Usher Be-718 Bookshelf Speakers with Beryllium Tweeters". Archived from the original on 13 June 2011. Retrieved 18 September 2008.
- ^ "Exposé E8B studio monitor". KRK Systems. Archived from the original on 10 April 2011. Retrieved 12 February 2009.
- ^ "Beryllium apply in pro audio Focal speakers". Archived from the original on 31 December 2012.
- ^ "VUE Audio announces use of Be in Pro Audio loudspeakers". VUE Audiotechnik. Archived from the original on 10 May 2012. Retrieved 21 May 2012.
- ^ Svilar, Marker (viii January 2004). "Analysis of "Glucinium" Speaker Dome and Cone Obtained from Communist china". Archived from the original on 17 May 2013. Retrieved 13 Feb 2009.
- ^ "Shure V15 VXmR User Guide" (PDF). Archived from the original (PDF) on 10 Jan 2017. Retrieved 31 May 2017.
- ^ Diehl, Roland (2000). Loftier-power diode lasers. Springer. p. 104. ISBN978-3-540-66693-6. Archived from the original on 27 July 2020. Retrieved 30 October 2021.
- ^ "Purdue engineers create safer, more efficient nuclear fuel, model its performance". Purdue University. 27 September 2005. Archived from the original on 27 May 2012. Retrieved eighteen September 2008.
- ^ Breslin AJ (1966). "Ch. iii. Exposures and Patterns of Disease in the Beryllium Manufacture". In Stokinger, HE (ed.). Beryllium: Its Industrial Hygiene Aspects. Bookish Press, New York. pp. 30–33. ISBN978-0126718508.
- ^ OSHA Hazard Information Bulletin HIB 02-04-19 (rev. 05-14-02) Preventing Adverse Wellness Effects From Exposure to Beryllium in Dental Laboratories
- ^ Elshahawy, W.; Watanabe, I. (2014). "Biocompatibility of dental alloys used in dental stock-still prosthodontics". Tanta Dental Periodical. 11 (ii): 150–159. doi:10.1016/j.tdj.2014.07.005.
- ^ "Beryllium 265063". Sigma-Aldrich. 24 July 2021. Archived from the original on 11 April 2021. Retrieved 21 December 2021.
- ^ a b c Emsley 2001, p. 57.
- ^ Venugopal, B. (xiv March 2013). Physiologic and Chemical Ground for Metal Toxicity. Springer. pp. 167–eight. ISBN9781468429527.
- ^ "Glucinium and Beryllium Compounds". IARC Monograph. Vol. 58. International Agency for Research on Cancer. 1993. Archived from the original on 31 July 2012. Retrieved 18 September 2008.
- ^ NIOSH Pocket Guide to Chemical Hazards. "#0054". National Institute for Occupational Safety and Health (NIOSH).
- ^ "CDC - NIOSH Pocket Guide to Chemical Hazards - Arsenic (inorganic compounds, as As)". Archived from the original on 11 May 2017. Retrieved 30 October 2021.
- ^ NIOSH Pocket Guide to Chemic Hazards - Mercury compounds. The National Institute for Occupational Safety and Wellness (NIOSH). Archived seven May 2021 at the Wayback Machine
- ^ a b "CDC – Beryllium Research- NIOSH Workplace Safety and Wellness Topic". www.cdc.gov. Archived from the original on xvi December 2016. Retrieved thirty Jan 2017.
- ^ Emsley 2001, p. five.
- ^ "Photo of Chicago Pile One Scientists 1946". Office of Public Affairs, Argonne National Laboratory. 19 June 2006. Archived from the original on xi Dec 2008. Retrieved 18 September 2008.
- ^ Newport News Shipbuilding Workers Face a Hidden Toxin Archived xiii January 2014 at the Wayback Machine, Daily Press (Virginia), Michael Welles Shapiro, 31 August 2013
- ^ International Program on Chemic Safety (1990). "Beryllium: Ecology Health CRITERIA 106". Earth Health Organisation. Archived from the original on ix June 2011. Retrieved ten Apr 2011.
- ^ "ASTM D7458 –08". American Society for Testing and Materials. Archived from the original on 12 July 2010. Retrieved 8 August 2009.
- ^ Minogue, E. M.; Ehler, D. S.; Burrell, A. K.; McCleskey, T. M.; Taylor, T. P. (2005). "Development of a New Fluorescence Method for the Detection of Beryllium on Surfaces". Journal of ASTM International. two (9): 13168. doi:x.1520/JAI13168.
- ^ "CDC – NIOSH Publications and Products – NIOSH Manual of Belittling Methods (2003–154) – Alpha List B". www.cdc.gov. Archived from the original on 16 December 2016. Retrieved xxx January 2017.
Cited sources [edit]
- Emsley, John (2001). Nature's Edifice Blocks: An A–Z Guide to the Elements . Oxford, England, UK: Oxford University Printing. ISBN978-0-19-850340-8.
- Mackay, Kenneth Malcolm; Mackay, Rosemary Ann; Henderson, W. (2002). Introduction to modern inorganic chemistry (sixth ed.). CRC Press. ISBN978-0-7487-6420-four.
- Weeks, Mary Elvira; Leichester, Henry 1000. (1968). Discovery of the Elements . Easton, PA: Journal of Chemic Education. LCCCN 68-15217.
Farther reading [edit]
- Newman LS (2003). "Beryllium". Chemical & Technology News. 81 (36): 38. doi:10.1021/cen-v081n036.p038.
- Mroz MM, Balkissoon R, Newman LS. "Beryllium". In: Bingham E, Cohrssen B, Powell C (eds.) Patty's Toxicology, Fifth Edition. New York: John Wiley & Sons 2001, 177–220.
- Walsh, KA, Glucinium Chemistry and Processing. Vidal, EE. et al. Eds. 2009, Materials Park, OH:ASM International.
- Glucinium Lymphocyte Proliferation Testing (BeLPT). DOE Specification 1142–2001. Washington, DC: U.Due south. Section of Energy, 2001.
External links [edit]
- ATSDR Example Studies in Environmental Medicine: Beryllium Toxicity U.S. Department of Wellness and Human being Services
- It'due south Elemental – Glucinium
- MSDS: ESPI Metals
- Beryllium at The Periodic Table of Videos (University of Nottingham)
- National Found for Occupational Safety and Health – Beryllium Page
- National Supplemental Screening Program (Oak Ridge Associated Universities)
- Historic Price of Beryllium in United states
How Many Protons In Beryllium,
Source: https://en.wikipedia.org/wiki/Beryllium
Posted by: eilermanfriltang83.blogspot.com

![{\displaystyle {\ce {[Be(H_2O)_4]^{2+}}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/8c300bc0a3682a741d256b1a6a8dcb246a855239)
![{\displaystyle {\ce {[Be(H_{2}O)_{4}]^{2+}{+}nF^{-}\leftrightharpoons Be[(H_{2}O)_{2-n}F_{n}]^{(2-n)\pm }{+}nH_{2}O}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/9a912c3571a62ed845ae0e535cb0a611b1b19518)
![{\displaystyle {\ce {[Be_3O(H_2PO_4)_6]^{2-}}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/4a5d9b088328a7ec7a57f65641f44ab8eab4e152)

![{\displaystyle {\ce {H_{2}A{+}[Be(H_{2}O)_{4}]^{2+}\leftrightharpoons [BeA(H_{2}O)_{2}]{+}2H^{+}{+}2H_{2}O}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/88afd13645c1b2eca86d7ba9ba8586abca494284)
![{\displaystyle {\ce {H_{2}A{+}[BeA(H_{2}O)_{2}]\leftrightharpoons [BeA_{2}]^{2-}{+}2H^{+}{+}2H_{2}O}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/a36992a15f44668eff511201715607bfbbfcdfaa)
![{\displaystyle {\ce {Na_4[Be_6(OCH_2(O)O)_6]}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/eb11d9d4fdbfc6972b29c3f0da2932c7af31f59a)


0 Response to "How Many Protons In Beryllium"
Post a Comment